COVID Vaccine for Children – Should You Vaccinate Your Kids?

Should I Vaccinate My Child Against COVID?
Covid Vaccine Safe For Children
Covid Vaccines Are Available For Children
Vaccines Not Yet Approved In Children
What Age Can I Vaccinate My Child
Covid Vaccine Have Side Effects In Children
Children Get A Booster COVID Shot
If I Don't Vaccinate My Child Against COVID-19
Updated 9th August 2022
This article is continuously updated.
The biggest motivation for getting your kids vaccinated is resuming normal life without the risk of getting infected.
With the world emerging from lockdown, children are more vulnerable to getting the coronavirus now more than ever. Although pediatric infections are usually not severe, children can still get sick, develop complications, and spread the virus.
So, if you have the opportunity, vaccinate your kids against COVID-19 to protect them and others around them.
Worldwide, pediatric COVID vaccines like Pfizer, Coronavac, and Sinopharm have been approved for children as young as 3 years old. These vaccines have undergone, and are still going through, extensive evaluation to make sure that they’re effective and safe for our kids.
To help you get started, here’s what you need to know when vaccinating your children against COVID-19.
Should I Vaccinate My Child Against COVID?
The short answer is yes. Health professionals and infectious disease experts worldwide believe that COVID-19 vaccination for your children is essential in strengthening their immune system and keeping them safe against severe disease. Since the pandemic began, over 8,000 children under the age of 11 have been hospitalized because of the virus, and almost 100 children have died.
There are many benefits to getting kids vaccinated for COVID-19:
- The vaccine helps prevent your child from getting COVID-19
- The vaccine reduces the transmission or spread of COVID-19 and protects the community
- The vaccine can help your child return to a normal life
- The vaccine prevents other Coronavirus variants from emerging
The Coronavirus can easily be transmitted among unvaccinated children, giving it more chances to infect others and for new variants to arise. With COVID-19 cases increasing in children, the vaccine is essential in protecting your child, and others, from getting the virus.
Is The COVID Vaccine Safe For Children?
Before any vaccine may be administered, it must undergo several vaccine trials and pass specific safety standards. The same goes for the COVID vaccine.
After thorough evaluation, the U.S. Food and Drug Administration (FDA) has determined the COVID-19 vaccine safe for children.
Data from a study conducted in the U.S., Spain, Finland, and Poland reported only mild side effects among the 4,600 children included in the study. The side effects were entirely typical after receiving a shot and went away within 2 days.
Another study conducted by Pfizer-BioNTech showed that the vaccine dosage for kids ages 5 to 11 is safe, well-tolerated, and effective in improving their immune response. The FDA also assessed the benefits and potential risks of the COVID-19 vaccine for kids 5 through 11 and concluded that the overall benefits outweigh the risks.
As of June 2022, the FDA has granted both the Pfizer-BioNTech and Moderna COVID-19 vaccines emergency use authorization in children as young as 6 months old.
Which Covid Vaccines Are Available For Children?
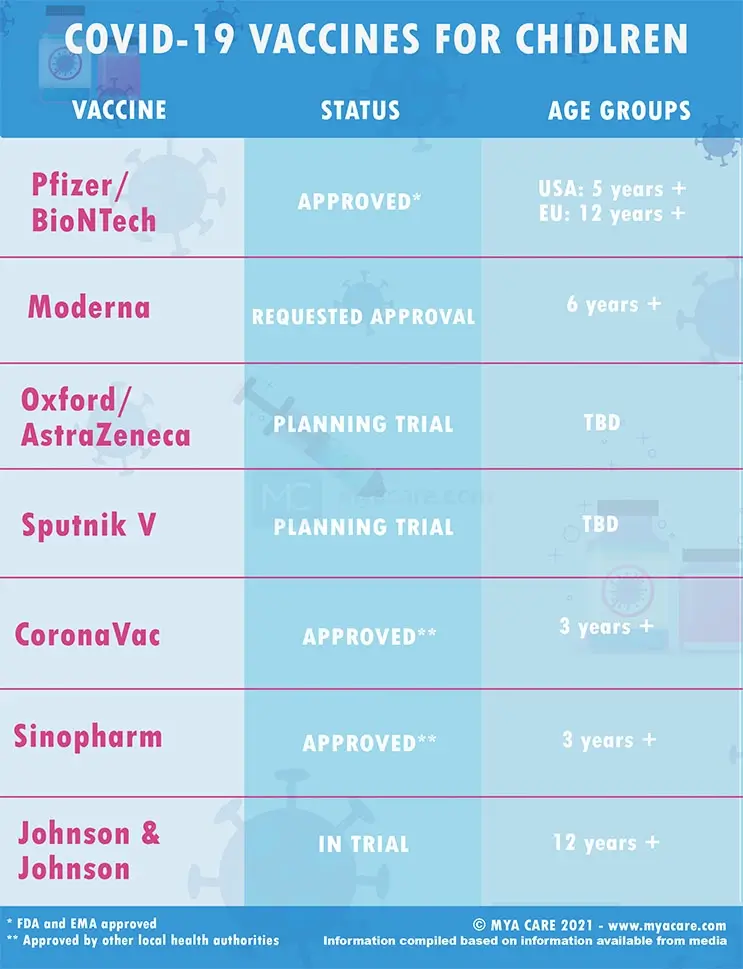
At the time of writing of this article, only 3 vaccines are approved for pediatric use: Pfizer/BioNTech, Coronavac, and Sinopharm. Other vaccines, like Moderna, Sputnik, AstraZeneca, and Johnson and Johnson are still being tested in children, but not yet approved.
Pfizer-BioNTech Vaccine
Is The Pfizer Vaccine Approved in Children?
The Pfizer-BioNTech COVID-19 vaccine has been approved for children in the United States, the European Union, and the UAE.
The U.S. FDA and UAE’s Ministry of Health and Prevention (MoHaP) have granted the emergency use authorization (EUA) of the Pfizer-BioNTech vaccine for children to prevent the spread and infection of COVID-19 in children ages 5 and up.
The European Union’s Medicines Agency (EMA) has also recently approved Pfizer/BioNTech for kids 5 and older in the EU.
More recently, the FDA approved the use of the Pfizer/BioNTech vaccine for kids between the ages of 6 months and 4 years.
Is The Pfizer Vaccine Safe In Children?
The FDA’s evaluation of the safety of Pfizer’s COVID-19 vaccine determined the vaccine safe for children aged 5 to 11. The most commonly reported minor side effects included pain at the injection site, fatigue, and headache, which usually resolve within 2 days after vaccination.
Data from the study showed that less than 0.2% of the kids experienced severe adverse effects. These adverse effects were common among this age group and considered unrelated to the specific vaccine. There were no reports of myocarditis (heart muscle inflammation).
July 29 2022 Update
The FDA’s evaluation of Pfizer’s COVID-19 vaccine safety in children determined the vaccine safe in individuals 6 months through 4 years of age. Some of the side effects that were reported include chills, nausea, fever, feeling unwell, and joint pain.
Severe side effects such as severe allergic reaction, myocarditis, and pericarditis were extremely rare.
Which Age Groups Can Get The Pfizer Vaccine?
Adolescents, ages 12 and older, can receive the Pfizer-BioNTech COVID-19 vaccine in the exact dosage as an adult, that’s 30 micrograms.
Children 5 through 11 years old can get a lower dose of the vaccine, 10 micrograms. Their vaccine is administered as a 2-dose series, delivered 3 weeks apart.
Children 6 months through 4 years can receive the Pfizer COVID vaccine as an injection into the muscle in 3 doses. The first 2 doses are given three weeks apart followed by a 3rd dose administered at least 8 weeks after the second dose.
Coronavac Vaccine
Is The Coronavac Vaccine Approved In Children?
Yes. Health regulators in several countries have approved the emergency use of the Coronavac or Sinovac vaccine for children ranging from 3 to 17 years old.
Local authorities in China, Cambodia, and Chile authorized the Coronavac vaccine for children to cut down the rising cases of COVID-19. Indonesia's and Malaysia's health ministry has also approved the Sinovac vaccine for kids. Although, Malaysia has only given conditional approval for adolescents with comorbidities and allergies.
Is The Coronavac Vaccine Safe In Children?
A randomized trial was conducted in healthy children and adolescents aged 3 to 17. Data from the trial shows that, out of 550 participants who received the Sinovac jab, 27% showed mild-to-moderate side effects. Less than 1% of the group showed severe adverse reactions that were not related to the vaccine.
Common side effects of the vaccine were injection-site pain and fever, and most children recovered within 2 days.
Researchers at the Hebei Provincial Centers for Disease Control and Prevention reported that the 3 microgram dose of the Coronavac vaccine for children was safe and well-tolerated.
Which Age Groups Can Get The Coronavac Vaccine?
Several countries have expanded the use of the Sinovac vaccine in children to reduce the spread of COVID-19.
The following age groups can receive the Sinovac vaccine:
- China: Ages 3 to 17
- Indonesia, Cambodia, Chile: Ages 6 to 17
- Malaysia: Ages 12 to 17 who have comorbidities, allergies, and cannot receive the Pfizer vaccine
Moderna Vaccine
Is The Moderna Vaccine Approved In Children?
Yes. The FDA has approved the use of the Moderna COVID-19 vaccine in individuals 6 months through 17 years of age.
The effectiveness of the Moderna vaccine was evaluated in two randomized clinical clinics which enrolled infants, children, and adolescents.
In all three age groups (6 months through 5 years, 6 through 11 years, and 12 through 17 years), the Moderna COVID-19 vaccine produced a strong immune response comparable to the immune response in adults.
Is The Moderna Vaccine Safe In Children?
The safety of the Moderna vaccine in children was evaluated in approximately 1,700 children 6 through 23 months, 2000 children 2 through 5 years, 3000 children 6 through 11 years, and 2500 individuals 12 through 17 years.
In individuals of ages 6 months through 5 years, the most commonly reported side effects were mild and included:
- Fever
- Pain
- Redness and swelling at the injection site
- Swelling
The most commonly reported side effects for individuals of ages 6 years through 17 years included:
- Pain
- Chills
- Fatigue
- Redness at the injection site
- Muscle and joint pains
More severe side effects such as a severe allergic reaction, myocarditis, pericarditis, and fainting were very rare.
Which Age Groups Can Get The Moderna Vaccine?
The FDA has approved the emergency use of the Moderna coronavirus vaccine in individuals 6 months through 17 years of age. The vaccine is to be administered in two doses one month apart.
The Moderna vaccine has already been authorized for use in adults 18 years and older.
Sinopharm Vaccine
Is The Sinopharm Vaccine Approved In Children?
Yes. After extensive trials, China’s National Biotec Group and the UAE’s National Crisis and Emergency Management Authority (NCEMA) have approved the Sinopharm vaccine for children.
Following their approval, health regulators in Cambodia and Argentina have also allowed the Sinopharm vaccine to immunize their children against COVID-19 to prepare for the re-entry of tourists.
Is The Sinopharm Vaccine Safe In Children?
Clinical trials conducted on 900 children, including members of Abu Dhabi’s ruling family, revealed that 96.6% of children developed a high antibody response against the Coronavirus. The study also found that the kids only received minor side effects after getting the 2 shots - 29% experienced injection-site pain, 8% experienced headache, and 3% had a fever.
After a thorough evaluation, the NCEMA concluded that the Sinopharm vaccine was safe and effective for children.
Which Age Groups Can Get The Sinopharm Vaccine?
Health regulators in the UAE, China, and Argentina have authorized the emergency use of the Sinopharm COVID-19 vaccine in children ages 3 to 17.
Local authorities in Cambodia have allowed children as young as 6 to 11 years old to get the Sinopharm shot.
Vaccines Not Yet Approved In Children
Aside from the vaccines mentioned above, several other companies have been conducting trials to test the efficacy and safety of their COVID vaccine for children.
Johnson & Johnson is studying its single-dose COVID-19 vaccine in adolescents aged 12 to 17. But, details of their trial have not yet been released or discussed by health officials. Oxford/AstraZeneca and Sputnik V also have plans to conduct vaccine trials for children under 17.
Moderna’s pediatric Coronavirus vaccine has shown that the vaccine is safe and produces a strong immune response in children ages 6 and up. The EMA has recommended using Moderna’s vaccine for children ages 12 to 17; however, it has yet to be approved by U.S. and European health regulators. The Moderna vaccine for kids shows promising results and just might be the next vaccine made available for children.
At What Age Can I Vaccinate My Child Against Coronavirus?
For over a year, vaccination against the COVID-19 has been going on worldwide. With the world going back to normal, vaccinating your kids against Corona is now more critical than ever.
Vaccine manufacturers have created pediatric doses of their COVID vaccines to administer to children to protect them against infection.
The pediatric COVID-19 vaccines can be administered to children as young as 5 years in the United States. In China, UAE, and Argentina, any child over 3 years old can get the shot, whereas, in Malaysia and the EU, they have to be at least 12 years old.
Once your local health officials authorize the use of the vaccine in children, they can receive the COVID vaccine together with their other routine vaccines.
Does The COVID Vaccine Have Side Effects In Children?
Like most vaccinations, the possible minor side effects of the COVID vaccine are typical signs that your child’s body is building immunity against the virus.
Your child may experience:
- Redness, swelling, and pain in the area where they received the shot
- Muscle pain
- Tiredness
- Headache
- Fever
- Chills
- Nausea
These symptoms are common effects of vaccines. They are temporary and should go away within 2 to 3 days.
One of the rare side effects that the FDA continually studied was myocarditis. Myocarditis is the inflammation of your heart muscles. Cases of myocarditis have been reported among children after receiving their second dose of the Pfizer-BioNTech COVID-19 vaccine.
The FDA and CDC safety systems observed a higher risk of myocarditis in males 12 to 17 years old, with roughly 11 cases for every 100,000 vaccinated males. There were no reported cases in children 5 to 11 years old.
Most kids with myocarditis respond well to medications and see a quick improvement in their symptoms.
The CDC states that the protection against COVID still outweighs the potential risk of a rare adverse effect to the vaccine and recommends that everyone have their child vaccinated.
Do Children Get A Booster COVID Shot?
There is currently no available data on whether children will need a booster shot of the COVID-19 vaccine.
The CDC has only given emergency authorization for a booster dose of COVID-19 vaccines in specific populations, none of which includes children (yet). The following groups can get a booster dose at 6 months or more after their initial doses:
- 65 years old and above
- Ages 18 and up who live in long-term care settings
- Ages 18 and up who have underlying medical conditions
- Ages 18 and up who work or live in high-risk settings
Up until the time of writing of this article, authorities have not yet recommended a third booster shot in children, regardless of whether or not they belong to higher-risk groups (immunocompromised or have a chronic medical condition).
What Happens If I Don't Vaccinate My Child Against COVID-19?
Allowing our kids to go on unvaccinated increases their risk of getting infected with COVID-19. They will be more likely to spread the disease to other children or adults, contributing to the formation of mutants that are resistant to available vaccines and treatments.
Although children infected with the Coronavirus usually have milder symptoms than adults, they can still become severely sick and get hospitalized if they’re not vaccinated.
If your child has an underlying medical condition, they are more at risk of developing severe COVID-19. They can also develop complications, such as Multisystem Inflammatory Syndrome (MIS-C), wherein their heart, lungs, kidneys, brain, skin, eyes, or gastrointestinal organs, can become inflamed.
Several private schools and daycare centers are calling for mandatory vaccination against COVID-19 to reduce the risk of spreading the virus. Vaccinating your children can help keep them in school, safely participate in activities, and give them a normal life.
Which Is The Best COVID Vaccine For Children?
Ultimately, the best COVID vaccine for children is any approved vaccine that’s available.
With the COVID vaccine, there is no number one. Manufacturers and health agencies have performed extensive clinical trials, reviews, evaluations, and other measures to ensure the efficacy and safety of the COVID-19 pediatric vaccine. Children are a vulnerable population, and no vaccine would be approved unless it has clearly and undoubtedly proven its safety and efficacy in this population.
If you’re still looking for the ‘best’ COVID vaccine for kids, you can talk to your healthcare provider and use this article as a guide to figure out which vaccine is suitable for your children.
With that being said, it’s a safe move to get your children vaccinated, no matter which vaccine. It doesn’t completely COVID-proof us against the virus, but it’s one step closer to living a healthy, normal life.
Frequently Asked Questions (FAQs)
What are the symptoms of myocarditis?
Myocarditis can manifest as chest pain, shortness of breath, and the feeling of having a pounding heartbeat. If you or your child experience any of these symptoms, especially a week after COVID-19 vaccination, seek medical care immediately.
Are there any side effects of the COVID-19 vaccine in children?
Side effects of the COVID-19 vaccine include pain or redness at the injection site, low-grade fever, headaches, muscle pain, chills, and nausea. However, these are commonly seen in most vaccinations and usually resolve in 2 days at most.
Can I get COVID-19 from my child?
Yes. If your child is infected with COVID-19, they can transmit the virus to another person, child or adult. COVID vaccines for kids lessen the risk of getting the virus from your child.
Does going back to school increase my child’s risk of catching the coronavirus?
The Delta variant has been highly contagious in unvaccinated people, including children. Safety precautions like indoor masking, physical distancing, and other measures are set in schools regardless of your child’s vaccination status. Having your children vaccinated as soon as possible significantly helps prevent infections and the spread of COVID.
Do schools require COVID vaccination for students?
Private schools, camps, and daycare centers may soon call for mandatory vaccination for their students, and some already have. Whether public K-12 schools will have vaccination requirements will depend entirely on your local authorities.
Can a child who recently received other vaccinations get the COVID shot?
Yes, the CDC says that Coronavirus vaccines can be given with other routine vaccines. If your pediatrician delivers multiple vaccines in one visit, they can provide the injections in different parts of the body.
Can I give my child pain relievers before getting the COVID-19 vaccine?
It is not recommended to give your child pain relievers, such as paracetamol, before vaccination to prevent side effects. However, you may give painkillers if your child develops side effects like fever or headache after the vaccination.
If you’re not already vaccinated, take the first chance you get. The best COVID-19 vaccine is the one that is available to you the soonest.
To search for Healthcare Providers in India providing COVID-19 Vaccine, please click here.
To search for Healthcare Providers in Malaysia providing COVID-19 Vaccine, please click here.
To search for Healthcare Providers in Poland providing COVID-19 Vaccine, please click here.
To search for Healthcare Providers in Thailand providing COVID-19 Vaccine, please click here.
To search for Healthcare Providers in UAE providing COVID-19 Vaccine, please click here.
To search for the best healthcare providers worldwide, please use the Mya Care search engine.
Dr. Mersad is a medical doctor, author, and editor based in Germany. He has managed to publish several research papers early in his career. He is passionate about spreading medical knowledge. Thus, he spends a big portion of his time writing educational articles for everyone to learn.
Featured Blogs